Research Areas
Our focus is in the following core research areas:
Joint Multi-Channel Infusion Pump
(JMCIP
— MP-1500)
NeuroWave is developing a new compact and rugged 3-channel large volume infusion pump in collaboration with the Air Force and other key DoD stakeholders.
The new pump is being developed as a replacement for the Medsystem III® device which has been recently discontinued by its manufacturer.
The pump has been tested in accordance with both the IEC 60601-2-24 standard and the newest AAMI TIR:101 (2021) technical reference in NeuroWave's pump testing laboratory. Designed to operate in any orientation, the pump provides an infusion and dosing accuracy profile that meets or exceeds that of commercial devices across its operational temperature, humidity and pressure range, and independently of backpressure, reservoir elevation and fluid viscosity. Its low size, weight and power footprint makes it an ideal candidate for future medical missions.
En-route & Prolonged Field Care Sedation (AutoSED)
NeuroWave has been working in collaboration with the US Navy to develop an infusion and sedation module for the Automated Critical Care System (ACCS), a small and compact under-the-litter platform designed to provide advanced critical care in the field. Our AutoSED™ platform attaches under a standard NATO litter and provides up to 4-channel infusion capability to the Navy's ACCS system. A miniature brain monitor mounted on the patient’s head provides constant and continuous information on the sedation level which can be used by medical professionals to adjust the drug delivery rate. Alternatively, the closed-loop controller embedded in the AutoSED system uses data from the brain monitor and can automatically maintain the patient's sedation level within the pre-defined range.
The AutoSED solution, and accompanying ACCS device, can also be used for prolonged field care to provide maintained sedation for stabilized casualties who are awaiting medical evacuation to higher echelons of care.
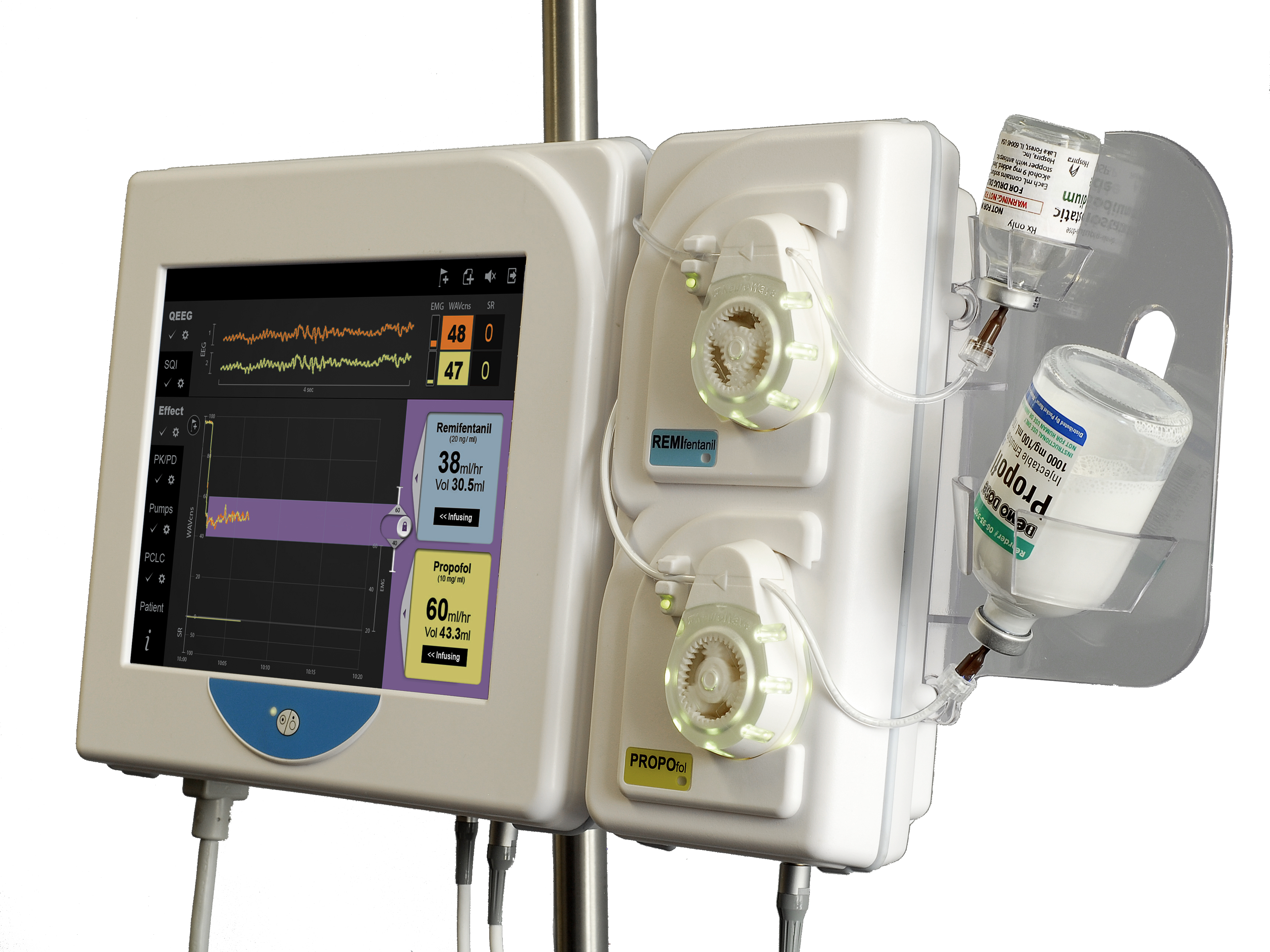
Anesthesia Auto-pilot (AutoTIVA)
NeuroWave is proud to introduce its anesthesia autopilot, AutoTIVA™ a device that enables care providers to deliver Total Intravenous Anesthesia (TIVA) in a safe and effective manner. Through automation, the delivery rate of propofol and remifentanil is continuously adjusted to maintain patients at the anesthesia depth targeted by the care provider. This system is powered by ongoing feedback from the NeuroWave NeuroSENSE monitor.
A 2-channel infusion pump module, based on a rotary peristaltic mechanism, attaches to the NeuroSENSE display monitor to create a compact and easy-to-use platform for drug delivery.
By combining automation, brain monitoring, and infusion technologies, the AutoTIVA enables care providers of all expertise levels to leverage the many benefits of TIVA for improved patient safety and outcome, while reducing the providers’ workload.